Here you will find the guidance and resources needed to obtain Ethics approval for your research proposal. Please review the College’s research policies to familiarize yourself with institutional and funding agency requirements before submitting your application.
Research Ethics Board (REB) mandate
- The mandate of the REB is to ensure that ethical principles are applied to all research involving human subjects.
- It has a role to educate researchers and to review and monitor research proposals and projects.
- It serves as a consultative body on research ethics and to assist in educating employees about research ethics.
- It has the responsibility for independent multidisciplinary review of research proposals to determine if they meet ethical requirements and to approve them to be initiated or continue.
Research policies at the College
All research activities associated with the College must receive appropriate College approval prior to commencement, i.e., no research or other study involving human subjects, animals, biological hazards or radioactive materials, or having any environmental impact may be undertaken unless approval has been given by the appropriate body: Research Ethics Board (human subjects), College Animal Care Committee (animals) or the Dean or Associate Dean, Science & Technology (biohazards, radioactive materials or environmental impact).
See list below for policies relevant to the conduct of research and scholarly activity at Douglas College (Administration Policies).
- Conflict of Interest
- Commercialization of Intellectual Property
- Integrity in Research and Scholarship
- Ethical Conduct for Research Involving Humans
- Records and Information Management
The Interagency Advisory Panel on Research Ethics (PRE) has developed an online introductory tutorial on the Tri-Council Policy Statement (TCPS 2). (Research involving humans.) It is self-administered and publicly available via PRE's website. For access to the tutorial, go to: https://tcps2core.ca/welcome
Contact the Office of the Vice President, Academic and Provost if you are engaging in the following kinds of research:
- Research/teaching with animals: You must submit your proposal to the Animal Care Committee for approval. Please review the Terms of Reference for the committee. The last page provides a framework for a submission to this committee.
- Research that has environmental impacts
- Research involving biohazards or radioactive material
Research Ethics Information and Process for Approval
The information below provides information on the process of research approval by the REB, as well as relevant resources for those interested in applying for approval, or wanting to learn more.
All research involving human subjects requires ethical review. The Cayuse Human Ethics platform accepts applications for the following types of research:
- Research conducted by Douglas College faculty.
- Research conducted by Douglas College faculty that has already been approved by another REB.
- Research projects conducted by Douglas College students under the supervision of a Douglas College faculty sponsor. (e.g., Honour Student research)
- Research conducted by an external Principal Investigator where a Douglas College faculty member is a Co-Investigator.
- Course-based research
- Research which is conducted by students under the supervision of an instructor as part of an approved course outline does not currently need approval from the Research Ethics Board, though this is a planned future requirement. Instructors do need to consider ethics when instructing students to “conduct research” in class including those items listed here.
- At present, the appropriate Faculty Education Committee will review the ethics of the generic research activities as part of its curricular review processes. The research activity must be listed in the course Curriculum Guidelines and must refer to the requirements laid out in this Policy. Faculty supervising students will ensure compliance with this Policy. Copies of appropriate generic consent forms and research ethics guidelines approved by the REB should be provided by the instructor to the students. In situations where student research activities will depart from using these forms, the faculty member should refer the matter to the REB for approval.
- Where students are carrying out research that is part of a faculty member's own research program, this research proposal must be reviewed by the REB.
- Research conducted by an external researcher who wishes to recruit Douglas College students or employees to participate in their research.
- Research conducted by Douglas College that meets the criteria for exemption from ethical review.
Please note that all Douglas College research team members (faculty and students) must have a Cayuse Human Ethics account. To access an account, please contact reb@douglascollege.ca External researchers who are not working with a Douglas College Co-Investigator can request a Guest Account. Please contact reb@douglascollege.ca.
Creating your account in the Human Ethics Platform.
If you do not yet have a Cayuse Human Ethics account, please contact reb@douglascollege.ca to request one. Once you have received a confirmation email that your account has been created, you can access your account by following this link: https://douglas.prod-ca.cayuse.com and signing in with your Douglas College credentials.
To help guide you in your Ethics application, please refer to the Cayuse Guide for Researchers manual that can be found here. Resources are also available on the REB DC Connect site for internal DC employees.
Please consult the checklist below to be sure that you have all the necessary information available for the application process.
Douglas College Research Ethics Board application checklist
If you are carrying out research as a faculty or student of Douglas College along with another REBC network institution, please see below under “Applying for a Harmonized Review”.
Information necessary for application on Cayuse
A draft of the Cayuse Human Ethics application form for your reference can be found here. Please see below for information that you will require.
- Copy of Tri-Council's on-line Course on Research Ethics (CORE) certificate
- Copy of human subject consent form.
- Copies of all questionnaires and/or test instruments
- Copies of human subject recruitment notices, emails, posters and/or advertisements.
- Copy of approval from other Research Ethics Boards (if applicable).
- Any additional information that researcher deems necessary for Research Ethics Board to view.
Research approval process
- The Applicant completes an initial submission through Cayuse Human Ethics platform.
- The REB completes an initial review of the application checking for completeness, level of risk and any initial information or revisions that are required. If revisions are needed, the application is returned to the applicant via Cayuse Human Ethics with comments.
- The applicant addresses the comments and resubmits the application.
- After initial review, if all issues have been sufficiently addressed, the application is forwarded for a delegated or full Board review, depending on the level of risk.
- The applicant will be notified by email of the REB decision.
- Approval is generally granted for one year, although duration of approval may be shorter where warranted. For research that continues past the one year approval, the Principal Investigator must request an extension by submitting an amendment request through Cayuse Human Ethics.
- Principal Investigators are required to report to the REB any and all significant unanticipated issues that arise with the research.
Please consult the College Policies on Research.
- The websites of the Tri-Council Funding agencies have a wealth of information related to the requirements for research and applying for funding:
- Introductory Tutorial for the Tri-Council Policy Statement (TCPS 2) on Ethical Conduct in Research with Humans: https://tcps2core.ca/welcome
Douglas College is now a member of the Research Ethics British Columbia (REBC) network and part of the province-wide, harmonized system for research ethics reviews of studies from more than one BC research institution. For an overview of REBC, click here.
If you are carrying out research as a faculty or student of Douglas College along with another REBC network institution, a harmonized application is required and should be submitted via the Provincial Research Ethics Platform or PREP. If any of the institutions are not members of REBC network or are outside of BC, researchers will be required to attain the approval from all institutions involved.
If you are unsure if your application requires harmonized review, please contact reb@douglascollege.ca. Applications requiring harmonized review will not be reviewed through the Douglas College application system. To learn more about the harmonized application process, please see below.
Application process
- PREP is available to Douglas College researchers and is hosted by UBC's Research Information System (RISe).
- To access PREP you need to obtain a UBC Campus Wide Login or CWL which can be done here: https://activate.id.ubc.ca/iamweb/
- For assistance follow these instructions: https://researchethicsbc.ca/apply-for-ethics-review/
- Douglas researchers can sign up for a basic account (i.e., non-UBC affiliated investigators).
- Once the CWL is created you will need to contact risesupport@ors.ubc.ca to register.
- RISe Support may contact Douglas to confirm your identity and requested permissions. This may take a day or two.
- Once you have an account and are registered, sign in to https://www.rise.ubc.ca/
Summary of REB application process
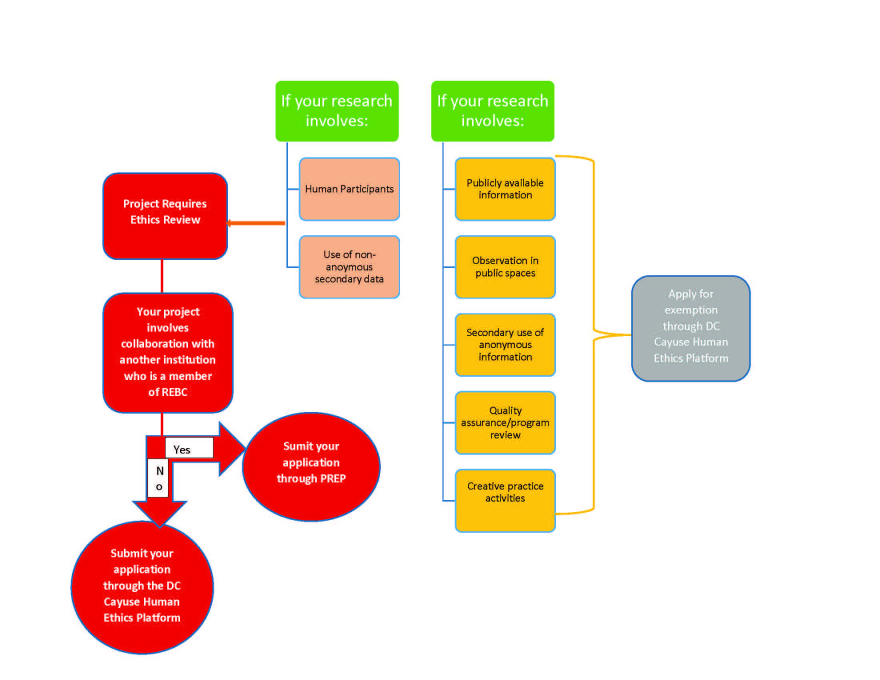
The REB has a mandate to ensure that ethical principles are applied to all research involving human participants. It educates researchers as well as reviews and monitors research proposals and projects. REB is responsible for independent review of research proposals to determine if they meet ethical requirements in order to get them approved, initiated or continued.
You need to submit an application through the Cayuse Human Ethics platform. Please see the “REB Application & Review Process” section above.
You will receive an email from the Research Ethics Board with an update to your status when approved.
If you are interested in research/teaching with animals you will need to contact the Office of the Vice President, Academic and Provost first. Then, you must submit your proposal to the Animal Care Committee for approval. Please review the Terms of Reference for the committee - the last page provides a framework for a submission to this committee.
When approved, you are free to move forward with your research as agreed upon by both parties.
Very few applications are rejected outright. It is more likely that revisions will be required before the REB can provide a certificate. It is commonplace for applications to have a few revisions. If your application is rejected, you can reapply after making the necessary changes to your methods as shared by the Research Ethics Board.
All research activities associated with the College must receive appropriate College approval prior to commencement. No research or other study involving human participants, animals, biological hazards, radioactive materials or research that has any environmental impact may be undertaken unless approval has been given by the appropriate body. The REB exists to ensure that all research conducted at the College meets the ethical requirements, as laid out here.
As the Provincial State of Emergency has been lifted, the DC REB is no longer operating under its Emergency Procedures and Directive related to research involving in-person activities. For in person research, please follow the Covid and Safe Return to Campus Resources and Information along with the current provincial health guidelines (https://www2.gov.bc.ca/gov/content/covid-19/info/restrictions).
The review process usually takes 3-4 weeks. Research involving more than minimal risk and requiring a full board review may take a little longer. When listing the intended start date for the research on your application make sure to consider these time frames.
“Expedited” is a term that is often used in the United States with the equivalent term being “delegated” in Canada. It does not mean fast-tracking. In some cases, for research that is time sensitive in nature or connected to an event, a quicker review can be requested. In general, researchers are encouraged to submit their application with the 3-4 week window in mind as well as time for any revisions that are requested by the REB. A comprehensive and well thought out ethics application can support a quicker review process.
All applications are evaluated for potential risk and vulnerability to participants. Delegated reviews are done for research that is deemed minimal risk. The review is completed by a subcommittee of the REB. Applications deemed higher risk are reviewed by the full board.
The online TCPS CORE Tutorial is an online self-paced educational program. The tutorial educates researchers around core ethical concerns in research involving human participants. All researchers (faculty, staff and students) are required to take the TCPS CORE Tutorial prior to submitting their ethics application. A certificate of completion for each researcher is to be submitted with the application to the REB.
Your research may involve multiple institutions that have their own ethical review processes. If the institutions are all in British Columbia, you may be eligible to submit your application for a harmonized review. Douglas College is a member of the Research Ethics British Columbia (REBC) network that provides a harmonized system for research ethics reviews of studies that take place at more than one BC research institution. If your research is in collaboration with another REBC network institution, a harmonized application is required. Please see the “Applying for Harmonized Review” section above. If any of the institutions are not members of REBC network or are outside of BC, researchers will be required to attain the approval from all institutions involved.
Updates on in-person research
As the Provincial State of Emergency has been lifted, the DC REB is no longer operating under its Emergency Procedures and Directive related to research involving in-person activities. For in person research, please follow the Covid and Safe Return To Campus Resources and Information along with the current provincial health guidelines.